
The reaction occurs when oppositely charged ions in solution overcome their attraction for water and bind to each other, forming a precipitate that separates out from the solution. 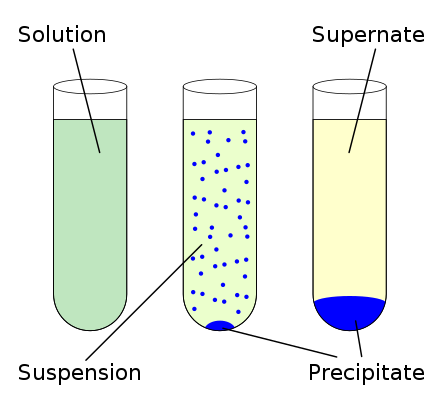
In a precipitation reaction, aqueous solutions of soluble salts react to give an insoluble ionic compound – the precipitate. In this reaction, sodium and silver cations exchange partners - pairing with nitrate and chloride anions, respectively - to form sodium nitrate and silver chloride. Salt metathesis is a chemical process in which the positive and negative ions of two ionic reactants exchange partners to form two new ionic products. This is an example of a salt metathesis reaction, which is also called a double displacement reaction. The molecular equation for this precipitation reaction suggests that aqueous sodium chloride reacts with aqueous silver nitrate to form aqueous sodium nitrate and solid silver chloride. Therefore, sodium nitrate is a soluble salt that remains in the solution as dissolved sodium ions and nitrate ions, while silver chloride is an insoluble salt that precipitates from the solution. 
Most chloride salts are water-soluble, but silver salt is an exception. According to this guide, all nitrate salts are water-soluble without exception. Referring to the solubility rules allows us to determine whether the ionic products are water-soluble or water-insoluble. Thus, sodium cations pair with nitrate anions to form sodium nitrate, and silver cations combine with chloride anions to form silver chloride. Since oppositely charged ions are attracted to each other, the cation from each reactant can pair with the anion from the other reactant to form new ionic products. When the two solutions are mixed, the resulting solution contains all four ions: the sodium and silver cations as well as the chloride and nitrate anions. Similarly, an aqueous solution of silver nitrate contains dissociated silver ions and nitrate ions. An aqueous solution of sodium chloride contains sodium ions and chloride ions in their dissociated forms. Sodium chloride and silver nitrate are water-soluble ionic compounds. The precipitate stays undissolved and can be separated from the solution by filtration. If mixing aqueous solutions of two different ionic compounds results in a chemical reaction that produces an insoluble ionic solid - called the precipitate - it is a precipitation reaction. Water molecules then surround the dissociated ions to form an aqueous solution. Water-insoluble ionic solids stay undissolved or undissociated in aqueous solutions. Water-soluble ionic solids dissolve by breaking up - or dissociating - into ions. 
This double replacement reaction will also produce aqueous sodium chloride.Ionic solids may be water-soluble or water-insoluble. Now, when you mix these two solutions, the calcium cations will pair up with the carbonate anions and for calcium carbonate, #"CaCO"_3#, an insoluble ionic compound that precipitates out of solution. Likewise, the calcium chloride solution will contain calcium cations, #"Ca"^(2+)#, and chloride anions, #"Cl"^(-)# You can thus say that the sodium carbonate solution will contain sodium cations, #"Na"^(+)#, and carbonate anions, #"CO"_3^(2-)# Sodium carbonate, #"Na"_2"CO"_3#, and calcium chloride, #"CaCl"_2#, are soluble ionic compounds that dissociate in aqueous solution to form cations and anions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
